|
On the other side of the periodic table elements gain electrons to resemble the next higher noble gas. Thus, the periodic table becomes a tool for remembering the charges on many ions. Thus, the group 2 metals tend to have a +2 charge. In many cases, elements that belong to the same group (vertical column) on the periodic table form ions with the same charge because they have the same number of valence electrons. 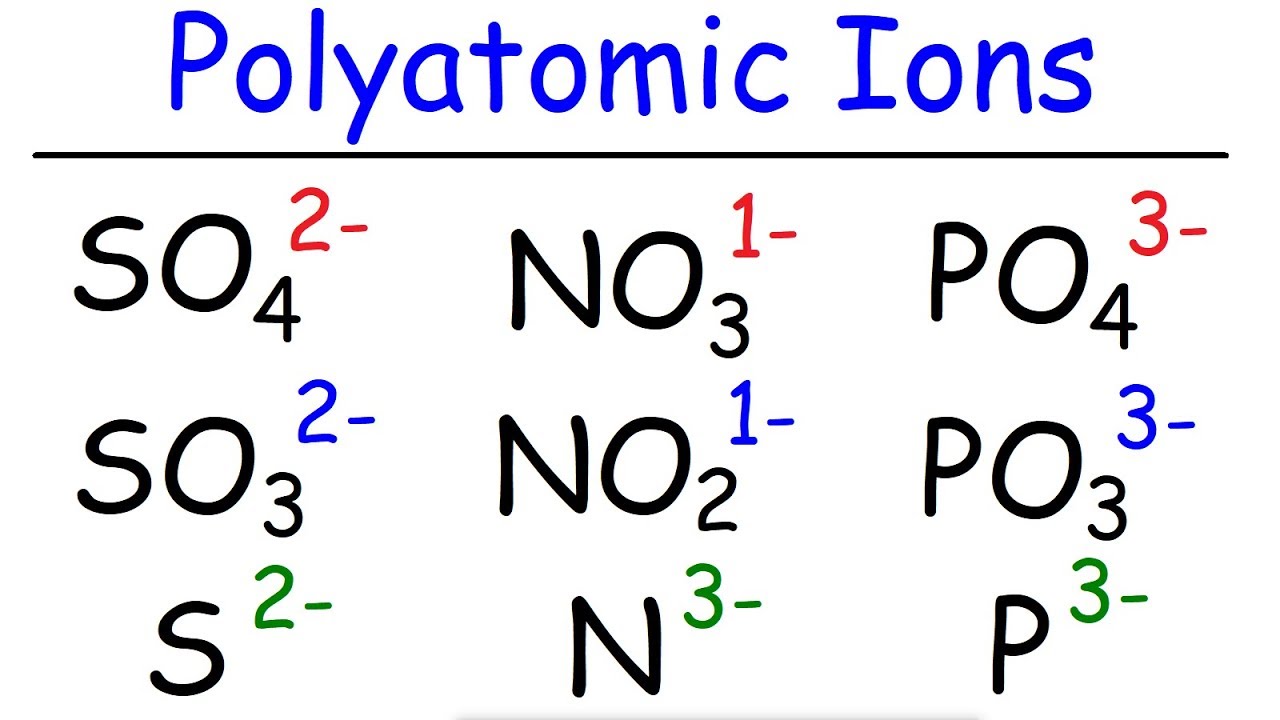
(a) The covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as Cl 2. The resulting anion is O 2 − with electron configuration, 1 s 22 s 22 p 6. Atomic radii are often measured in angstroms (Å), a non-SI unit: 1 Å 1 × 1010 m 100 pm. The second shell has six electrons ( 2 s 22 p 4) and needs two electrons to achieve octet. The electron configuration of O atom is 1 s 22 s 22 p 4. Use the periodic table to determine the charge in an element The overall charge in an ionic compound must be neutral All the negative charges must cancel out. The Periodic Table of Elements Provides Information on Ionic Charges. How many electrons must O lose/gain to achieve octet? Write the formula of the resulting ion and its electron configuration. The barium cation is written Ba 2+, not Ba +2. Note the convention of first writing the number and then the sign on a multiply charged ion. iron, silver, nickel), whilst most other nonmetals typically form. 3 shows how the charge on many ions can be predicted by the location of an element on the periodic table. Halogens always form anions, alkali metals and alkaline earth metals always form cations. Write the electron configuration of oxygen atom (Z=8). It can be possible to predict whether an atom will form a cation or an anion based on its position on the periodic table. In macroscopic samples of sodium chloride, there are billions and billions of sodium and chloride ions, although there is always the same number of cations and anions. The number of electrons lost by the sodium atom (one) equals the number of electrons gained by the chlorine atom (one), so the compound is electrically neutral. Notice that there are no leftover electrons. The resulting combination is the compound sodium chloride. With two oppositely charged ions, there is an electrostatic attraction between them because opposite charges attract. The ionic charge trend in the periodic table is that generally elements on the left form cations and those on the right form anions. On the right, the chloride ion has 18 electrons and has a 1− charge. The transition metals form similar compounds such as FeCl 3, HgI 2, or Cd. Mg 3 N 2, and CaS) in which there are just enough negative ions to balance the charge on the positive ions. 
On the left, the chlorine atom has 17 electrons. The elements in the periodic table are often divided into four categories: (1) main group elements, (2) transition metals, (3) lanthanides, and (4) actinides.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
